Varroa 3
Monitoring for Varroa in August/September - an 8-year experiment
Immediately after the honey supers are taken off a varroa screen is placed under each OMF for 4 days so that an idea of the natural mite drop can be found.
Thereafter each colony is treated with formic acid as described in Varroa 2 in the menu above.
Some results are given for the last 8 years in the charts.
Thereafter each colony is treated with formic acid as described in Varroa 2 in the menu above.
Some results are given for the last 8 years in the charts.
What conclusions can be drawn from these experiments over an 8-year eriod ?
1. Looking at the mite drop before treatment with formic acid generally gives little indication of the mite infestation.
2. It goes without saying that once formic acid is applied there is an immediate mite fall. The first count after 3 days is usually the greatest.
3. The mite drop in each 3 day period tends to decrease with each dose.
4. All these colonies were of a similar strength and history but there is a significant difference in the mite drop between colonies.
5. No detrimental effect was notice on the bees. Only when the formic acid was first applied did the bees set up a vigorous fanning action which quickly dispersed the fumes throughout the brood box. It was surprising to see that the bees did not avoid the pad as might be expected if they found the acid not to their liking!
What is important is that no evidence has been found which would suggest that the formic acid treatment as described has damaged a queen in any way.
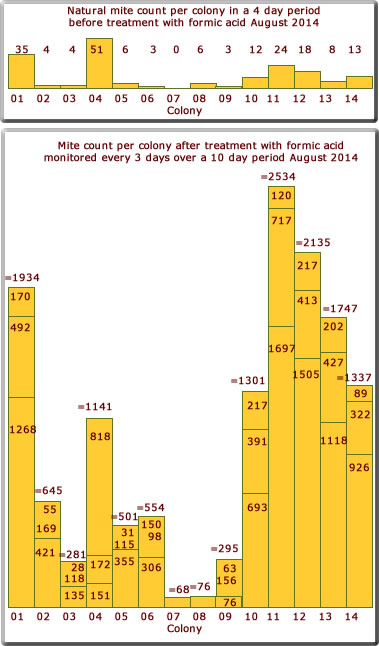
August 2014
The results for 14 colonies treated with formic acid in August 2014 are shown on the bar chart on the right (and some of the dead mites in the photo below).
What was very surprising this year was the relatively very high mite count compared to the two previous years.
The natural mite drop before the formic acid treatment gives little indication of the number of mites present per colony.
It is somewhat difficult to find an explanation for the sudden increase in the mite population this summer compared to previous summers. Perhaps the very good summer weather might be the cause allowing the mites to have more brood cycles.
In hindsight perhaps the colonies should have been treated with formic acid in the early Spring before the mite population had a chance to take off.
1. Looking at the mite drop before treatment with formic acid generally gives little indication of the mite infestation.
2. It goes without saying that once formic acid is applied there is an immediate mite fall. The first count after 3 days is usually the greatest.
3. The mite drop in each 3 day period tends to decrease with each dose.
4. All these colonies were of a similar strength and history but there is a significant difference in the mite drop between colonies.
5. No detrimental effect was notice on the bees. Only when the formic acid was first applied did the bees set up a vigorous fanning action which quickly dispersed the fumes throughout the brood box. It was surprising to see that the bees did not avoid the pad as might be expected if they found the acid not to their liking!
What is important is that no evidence has been found which would suggest that the formic acid treatment as described has damaged a queen in any way.
August 2014
The results for 14 colonies treated with formic acid in August 2014 are shown on the bar chart on the right (and some of the dead mites in the photo below).
What was very surprising this year was the relatively very high mite count compared to the two previous years.
The natural mite drop before the formic acid treatment gives little indication of the number of mites present per colony.
It is somewhat difficult to find an explanation for the sudden increase in the mite population this summer compared to previous summers. Perhaps the very good summer weather might be the cause allowing the mites to have more brood cycles.
In hindsight perhaps the colonies should have been treated with formic acid in the early Spring before the mite population had a chance to take off.
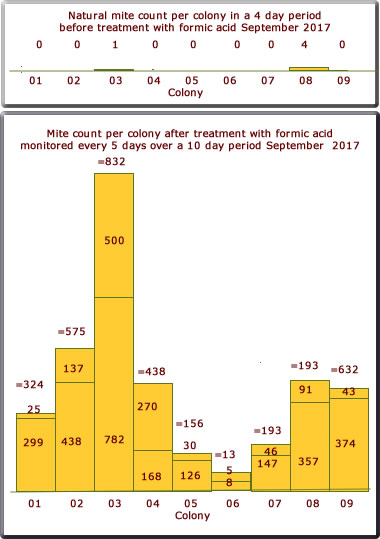
September 2017
The mite count is shown on the bar chart below. This is not to the same scale as the charts above. Only 9 colonies this year after scaling down a little.
The results were somewhat pleasing; only colony 03 had a significant mite drop.
Only two counts were possible this year over the 10 day period.
The mite count is shown on the bar chart below. This is not to the same scale as the charts above. Only 9 colonies this year after scaling down a little.
The results were somewhat pleasing; only colony 03 had a significant mite drop.
Only two counts were possible this year over the 10 day period.

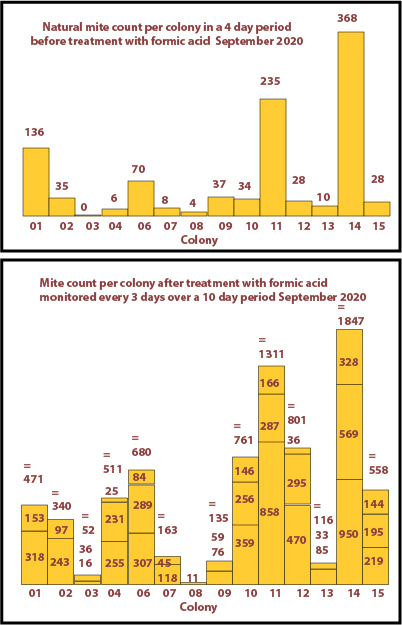
September 2020
14 colonies this year. The mite counts are shown on the bar charts below but not drawn to the same scale as those fot 2019.
Surprisingly there was a significant natural mite drop from most colonies.
After the treatment with formic acid varroa was seen to be present in most colonies. Only 2 doses of formic acid were given to those colonies with small mite counts.
14 colonies this year. The mite counts are shown on the bar charts below but not drawn to the same scale as those fot 2019.
Surprisingly there was a significant natural mite drop from most colonies.
After the treatment with formic acid varroa was seen to be present in most colonies. Only 2 doses of formic acid were given to those colonies with small mite counts.
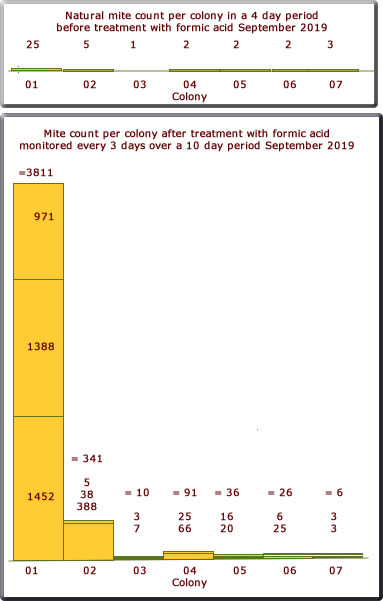
September 2019
The mite count is shown on the bar chart below. This is not to the same scale as the charts for 2017/19. Only 7 colonies this year.
Once again a surprising result only colony 01 and 02 had a significant mite drop; very little from the other colonies. But colony 01 was extraordinary with a record mite drop of 3811.
Only two applications for formic acid were used for colonies 03 -07 owing to the low mite count.
The mite count is shown on the bar chart below. This is not to the same scale as the charts for 2017/19. Only 7 colonies this year.
Once again a surprising result only colony 01 and 02 had a significant mite drop; very little from the other colonies. But colony 01 was extraordinary with a record mite drop of 3811.
Only two applications for formic acid were used for colonies 03 -07 owing to the low mite count.
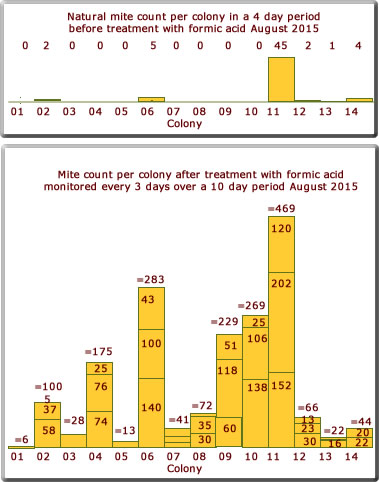
August 2015
The mite drop count shown in the bar charts to the right is not to the same scale as that above.
What is surprising is the relative low mite count this year compared to 2014. The weather perhaps provides the answer. Whereas 2014 was a very good summer, 2015 was quite poor with the mean temperature about 1.5 ℃ below the mean.
The relatively low mite count for some colonies (below 100) was very pleasing.
The mite drop count shown in the bar charts to the right is not to the same scale as that above.
What is surprising is the relative low mite count this year compared to 2014. The weather perhaps provides the answer. Whereas 2014 was a very good summer, 2015 was quite poor with the mean temperature about 1.5 ℃ below the mean.
The relatively low mite count for some colonies (below 100) was very pleasing.
September 2018
The mite count for 8 colonies is shown below to the same scale as for 2017. The results are a complete surprise! considering the few mites that were recorded.
In view of the fact that the mite count at this time of the year in past seasons has been sometimes over a 1000, this year results are extraordinary. Especially so since no specific mite control was performed during the season.
Do the results mean the bees are learning to control the mites. It will be interesting to see if there is a significant mite count in December when the colonies are treated with oxalic acid.
The mite count for 8 colonies is shown below to the same scale as for 2017. The results are a complete surprise! considering the few mites that were recorded.
In view of the fact that the mite count at this time of the year in past seasons has been sometimes over a 1000, this year results are extraordinary. Especially so since no specific mite control was performed during the season.
Do the results mean the bees are learning to control the mites. It will be interesting to see if there is a significant mite count in December when the colonies are treated with oxalic acid.

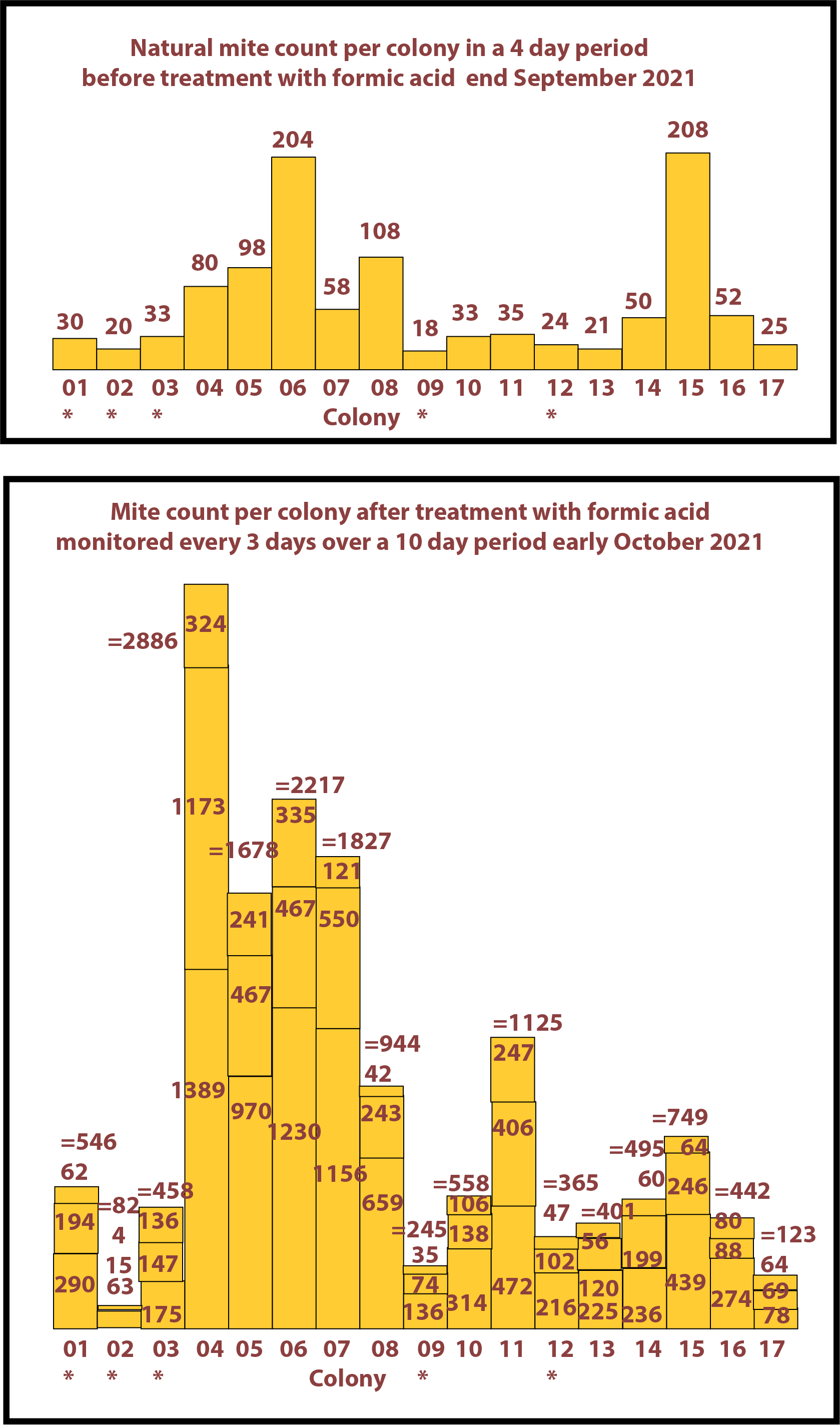
September 2021
17 colonies this year going into winter. Those colonies marked with (*) are small colonies building up from nucs. The mite counts are shown on the bar charts on the right. There was a significant natural mite count from all colonies.
After the treatment with formic acid a large amount of varroa was seen to be present in most of the main colonies.
17 colonies this year going into winter. Those colonies marked with (*) are small colonies building up from nucs. The mite counts are shown on the bar charts on the right. There was a significant natural mite count from all colonies.
After the treatment with formic acid a large amount of varroa was seen to be present in most of the main colonies.